Hundreds Of US Businesses Offer Unapproved Stem Cell Treatments: Why You Should Avoid Them

For years, companies abroad marketed stem cells as the cure for a variety of ailments, then sold these treatments at a hefty price. Known as stem cell tourism, the market consisted of illegitimate clinics that offered unapproved and unregulated therapies, with unknown benefits to the consumer. And while people mainly traveled outside the United States to receive these treatments, a new report finds that more and more of these clinics are operating inside the U.S.
In the report, bioethicist Leigh Turner, an associate professor at the University of Minnesota, teamed up with stem cell researcher Paul Knoepfler, a professor at UC Davis, to examine the culpable companies. They found that some 351 companies, across 570 clinics in the U.S., are selling bogus stem cell therapies to treat diseases from heart problems to neurological disorders.
“In almost every state now, people can go locally to get stem cell ‘treatments,’” said Knoepfler in a press release. “Many people in larger metropolitan areas can just drive 15 minutes to find a clinic offering these kinds of services instead of, say, traveling to Mexico or the Caribbean. I think this reflects a change from what we’ve seen documented in the past and is different from what we typically think about when we think of stem cell tourism.”
In order to track down these businesses, the researchers combed through Internet key word searches, text mining, and company websites. They recorded each company’s name, location(s), website, advertised treatments, and marketing claims, aiming to create a database that could help consumers avoid bogus treatments.
They found that certain states tended to feature the most clinics: California, Florida, Texas, Colorado, Arizona, and New York contained the most. Top cities included Beverly Hills, New York, San Antonio, Los Angeles, Austin, Scottsdale, and Phoenix. These businesses market treatments for orthopedic issues, sports injuries, heart problems, neurological diseases, and immune disorders.
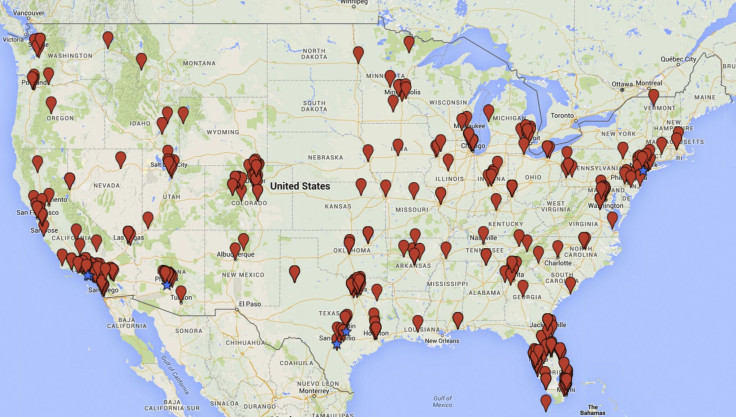
A New Type Of Stem Cell Tourism
“This is a marketplace that is dramatically expanding before our eyes — we were aware early on and tracked it early on, but I don’t think we knew the scope and size of the market,” said Turner in the press release. “Brakes ought to exist in a marketplace like this, but where are the brakes? Where are the regulatory bodies? And how did this entire industry come into being in a country where stem cell-based interventions and the medical devices that produce them are supposed to be regulated by the FDA?”
The researchers note that the businesses have been growing in the U.S. since 2009, yet the FDA or other authorities haven’t taken “meaningful regulatory action.” That leaves the researchers with the question: "Does that mean that people are getting access to safe and efficacious interventions or is there basically unapproved human experimentation taking place where people are going to these businesses and receiving experimental investigational cell-based interventions without being given a meaningful account of the lack of knowledge and evidence that they're being charged for?” Turner asks.
Last year, the FDA released draft guidelines that stated any stem cell treatments are drugs and must be closely regulated before they can be deemed safe. There is only one stem cell product that’s been approved by the FDA; known as Hemacord, it’s a cord blood-derived product produced by the New York Blood Center. Most other products or procedures being marketed — including fat-derived stem cell interventions or bone marrow-based treatments, which are pretty popular — are technically unapproved.
Source: Turner L, Knoepfler P, et al. “Selling Stem Cells in the USA: Assessing the Direct-to-Consumer Industry.” Cell Stem Cell, 2016.



























